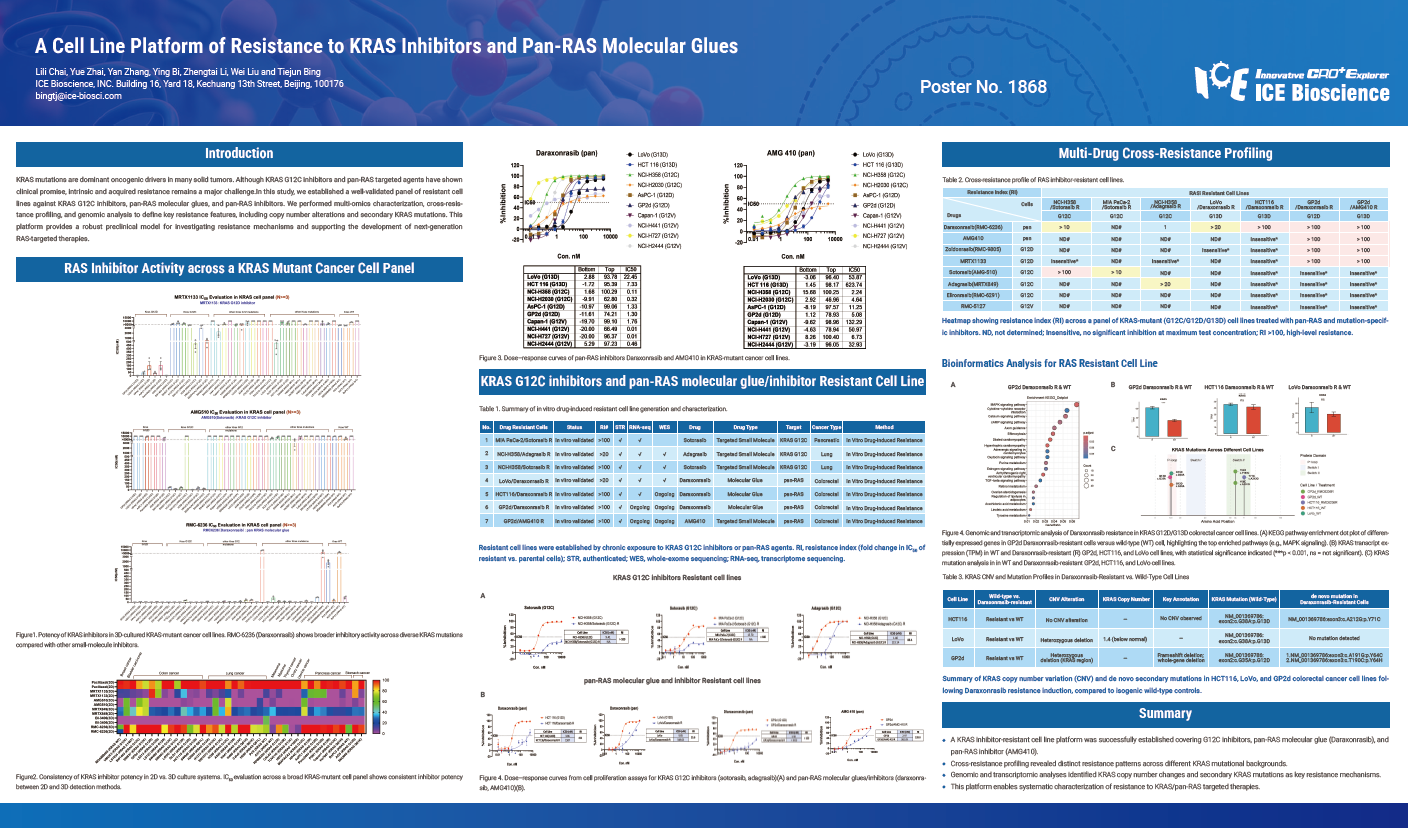
KRAS mutations are dominant oncogenic drivers in many solid tumors. Although KRAS G12C inhibitors and pan-RAS targeted agents have shown clinical promise, intrinsic and acquired resistance remains a major challenge.In this study, we established a well-validated panel of resistant cell lines against KRAS G12C inhibitors, pan-RAS molecular glues, and pan-RAS inhibitors. We performed multi-omics characterization, cross-resistance profiling, and genomic analysis to define key resistance features, including copy number alterations and secondary KRAS mutations. This platform provides a robust preclinical model for investigating resistance mechanisms and supporting the development of next-generation RAS-targeted therapies.

Cardiac safety liability assessment is essential in drug development to prevent arrhythmias like torsades de pointes (TdP), which can cause sudden death. Traditional ICH S7B and E14 guidelines focus on hERG blockade and QT prolongation, but these lack specificity, leading to high attrition or missed risks. The Comprehensive in vitro Proarrhythmia Assay (CiPA), launched in 2013 by FDA, academia, and industry, shifts to a mechanistic paradigm. It integrates in vitro ion channel assays, in silico modeling, hiPSC-CM evaluations, and clinical ECGs to predict proarrhythmia accurately, reducing animal use and improving efficiency. Validated with 28 reference compounds, CiPA enhances specificity.

In drug discovery, proactive toxicological risk assessment of candidate compounds is crucial for avoiding clinical-stage failure and post-market withdrawal. However, traditional preclinical safety evaluation strategies have significant limitations: in vivo animal studies exhibit low human relevance (translatability) due to significant species differences and face dual pressures of ethics and cost; while conventional in vitro binding assays are inadequate for predicting functional biological effects. Notably, approximately 75% of clinical adverse drug reactions (ADRs) originate from dose-dependent off-target effects, highlighting an urgent need for more accurate early-risk identification tools. To address these challenges, the IQ DruSafe consortium—a preeminent alliance of global pharmaceutical companies—has championed the enhancement of preclinical predictive power through the expansion and refinement of secondary pharmacology screening strategies. This study is aligned with this initiative and aims to investigate the implementation of an advanced in vitro secondary pharmacology screening system utilizing functional assay formats. By generating richer pharmacological information beyond mere binding affinity, this strategy seeks to enable a more accurate and earlier identification of potential safety liabilities, thereby providing highly translatable safety insights for the optimization of lead compounds.
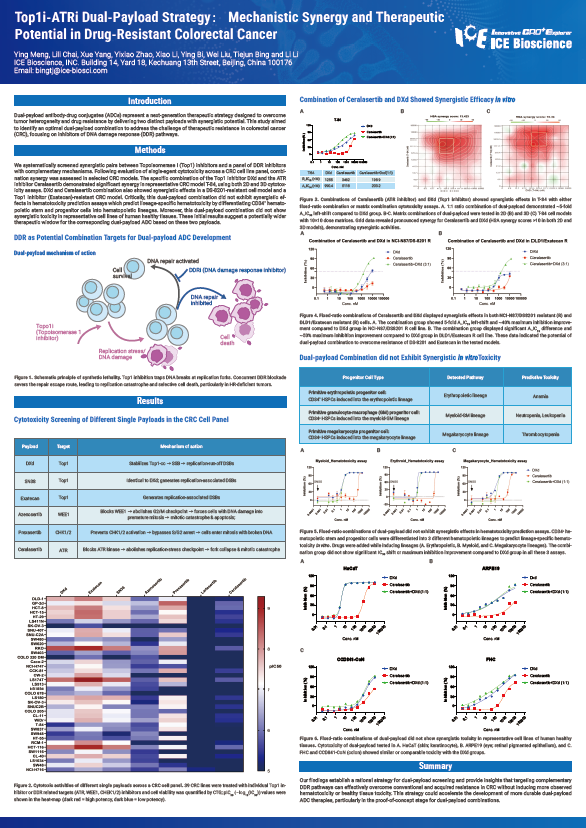
Dual-payload antibody-drug conjugates (ADCs) represent a next-generation therapeutic strategy designed to overcome tumor heterogeneity and drug resistance by delivering two distinct payloads with synergistic potential. This study aimed to identify an optimal dual-payload combination to address the challenge of therapeutic resistance in colorectal cancer (CRC), focusing on inhibitors of DNA damage response (DDR) pathways.