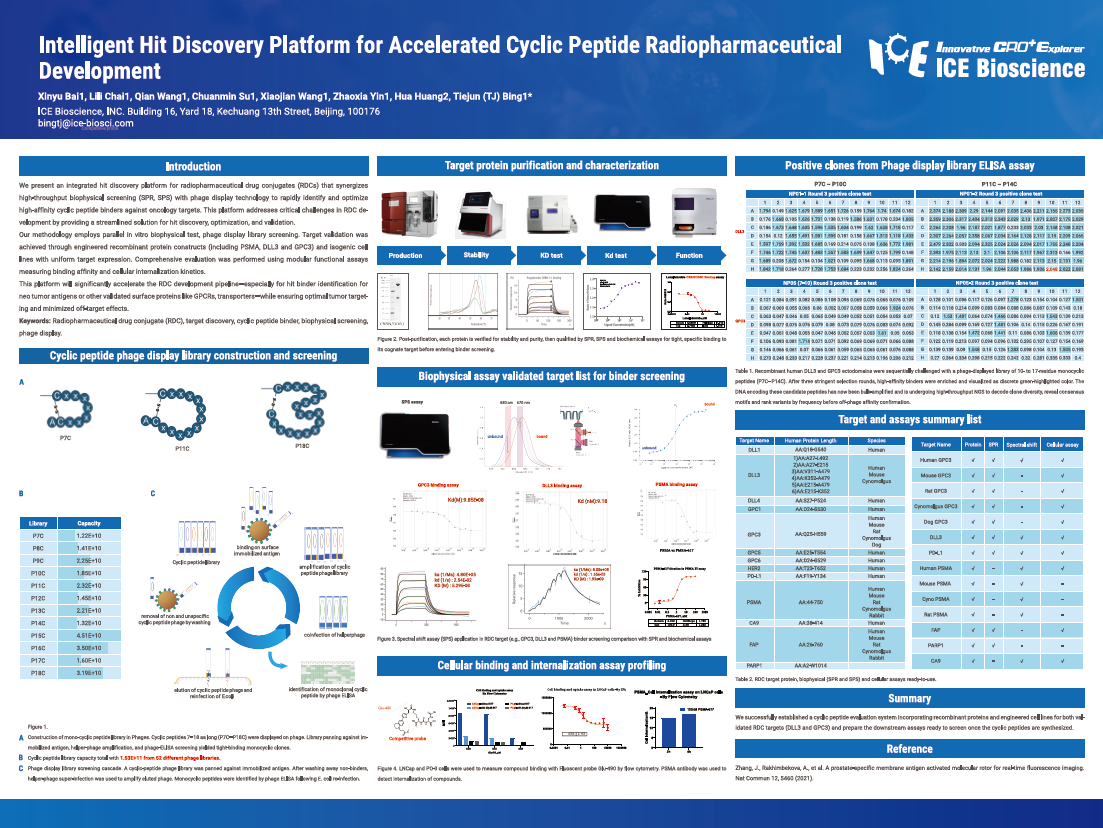
We present an integrated hit discovery platform for radiopharmaceutical drug conjugates (RDCs) that synergizes high-throughput biophysical screening (SPR, SPS) with phage display technology to rapidly identify and optimize high-affinity cyclic peptide binders against oncology targets. This platform addresses critical challenges in RDC development by providing a streamlined solution for hit discovery, optimization, and validation.
Our methodology employs parallel in vitro biophysical test, phage display library screening. Target validation was achieved through engineered recombinant protein constructs (including PSMA, DLL3 and GPC3) and isogenic cell lines with uniform target expression. Comprehensive evaluation was performed using modular functional assays measuring binding affinity and cellular internalization kinetics.
This platform will significantly accelerate the RDC development pipeline—especially for hit binder identification for neo tumor antigens or other validated surface proteins like GPCRs, transporters—while ensuring optimal tumor targeting and minimized off-target effects.
Early assessment of candidate drug safety is critical in pharmaceutical development to mitigate post-market withdrawals and reduce potential risks. Approximately 75% of Adverse Drug Reactions (ADRs) are dose-dependent (Type A), often mediated by interactions with unintended off-target proteins. Traditional binding assays have limitations in predicting functional biological effects. To address this challenge, this study aims to investigate how an in vitro secondary pharmacology screening strategy, utilizing a functional assay format, can enable more accurate identification of potential safety liabilities early in drug discovery by providing richer pharmacological data.
The characterization of biomolecular interactions is fundamental to drug discovery, particularly for complex modalities such as small molecules, molecular glues (MG), and PROTACs. This study presents a comparative analysis of several key biophysical technologies, with a focus on the innovative DIANTHUS platform.
DIANTHUS combines Spectral Shift-based detection with Time-Resolved Immuno-FRET (TRIC) to deliver superior performance in homogeneous assays. Compared to established techniques like HTRF and SPR, DIANTHUS demonstrates exceptional sensitivity, a broad dynamic range, and remarkable resilience to compound interference. These attributes make it an ideal solution for challenging applications, including the high-throughput screening and precise mechanistic study of molecular glues and PROTACs, which function by inducing or stabilizing protein-protein interactions. Our findings position the DIANTHUS platform as a powerful and versatile tool for accelerating the development of next-generation therapeutic agents.
Degrader-antibody conjugates (DACs) typically consist of a tumor-targeting antibody and a conjugated degrader payload, linked via a chemical linker. By integrating the degrader payload’s capacity to precisely degrade disease-causing proteins with the antibody’s tumor-targeting specificity, DACs emerge as an innovative and promising therapeutic modality for cancer treatment. Herein, we present the well-established DAC integrated platform developed by ICE Bioscience, which facilitates the characterization and evaluation of DACs from in vitro assays to in vivo studies.