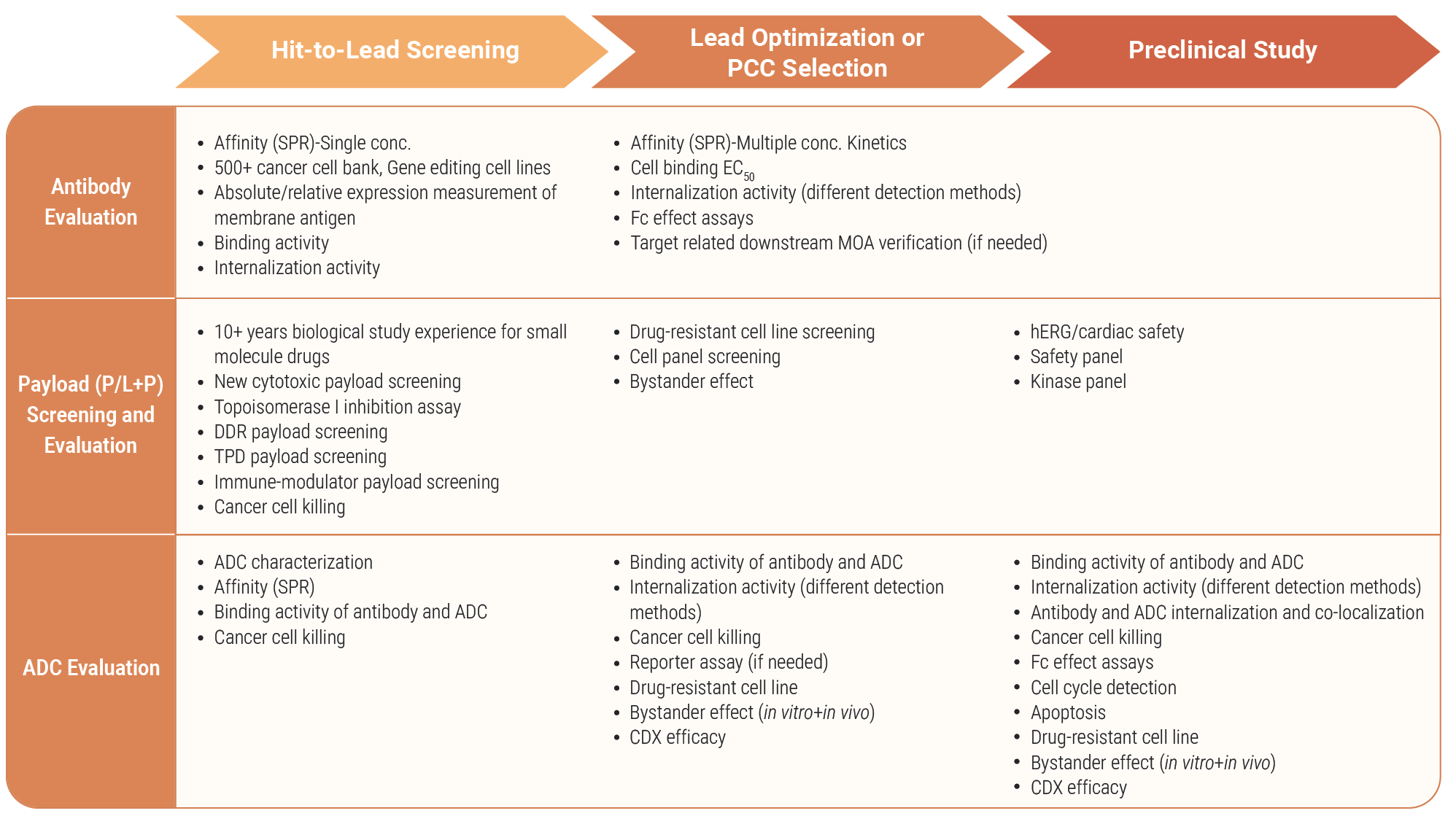
Our services are critical for optimizing the design and functionality of Antibody-Drug Conjugates (ADCs). These services provide comprehensive insights into ADC targeting, internalization, cytotoxicity, and stability, ensuring that each ADC is fine-tuned for maximum therapeutic efficacy and safety. By addressing key factors such as binding specificity, payload delivery, and drug stability, we help accelerate the development of highly effective ADCs for targeted cancer therapies.
Target-Specific Binding and Characterization: We ensure precise targeting by evaluating antigen expression, binding affinity, and antibody specificity, minimizing off-target effects.
Internalization and Payload Delivery: We assess ADC internalization efficiency to confirm that the payload is effectively delivered into target cells for maximum therapeutic impact.
Cytotoxicity and Mechanism of Action: Our assays measure the cytotoxic potency of the ADC, providing insights into how effectively it kills target cells while limiting damage to healthy cells.
ADC Design Optimization and Stability: We evaluate the stability and drug-to-antibody ratio (DAR) to ensure the ADC maintains its efficacy and integrity throughout its lifecycle.
Our live cell imaging-based cytotoxicity assay service provides real-time, kinetic analysis of cell viability and cytotoxic effects of therapeutic agents, including ADCs. Using the Incucyte live-cell imaging system, we offer a detailed and dynamic view of how your ADC affect cancer cell populations over time. Contact us for more detailed reference data.
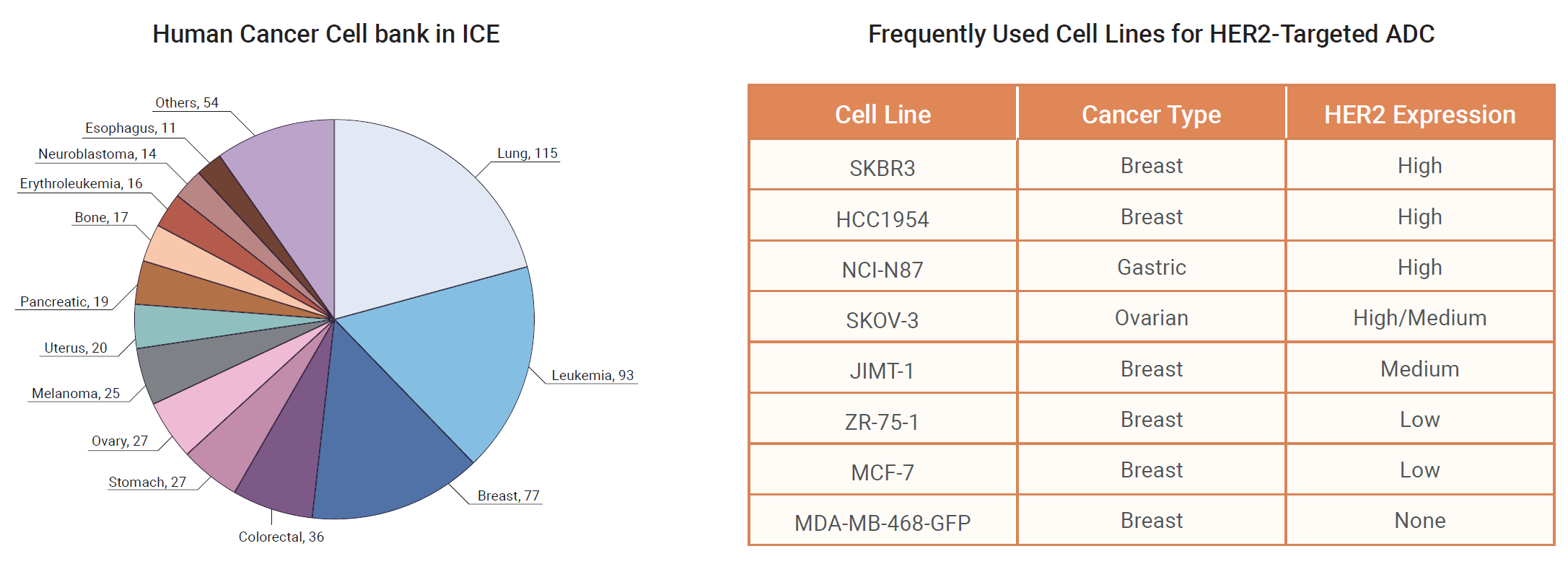
ADC Cancer Cell Panel is specifically designed for evaluating the efficacy of ADCs for specific targets using cell viability assays. For different ADC targets, we may supply tumor cell lines of different cancer types with different expression levels. In addition, drug-resistant cancer cell line models are available to support ADC resistance profiling and payload mechanism studies. Learn more.
Our in vitro bystander effect assay service is designed to evaluate payload diffusion and secondary cytotoxic effects beyond target-positive cells, a key mechanism underlying the efficacy and safety profile of ADCs. Using co-culture systems with defined target-positive and target-negative cancer cell populations, we assess payload-mediated bystander killing under controlled in vitro conditions, supporting payload selection, linker optimization, and mechanism-of-action studies. Learn more.
We provide comprehensive ADC conjugation and characterization services, including Size-Exclusion Chromatography (SEC) for analyzing ADC size and aggregation state, Hydrophobic Interaction Chromatography (HIC) for characterizing ADC hydrophobicity and conjugation status, and free payload/linker-payload (LP) detection for identifying and quantifying unbound payloads.
DAR Analysis (Drug-to-Antibody Ratio) quantifies the average number of drug molecules conjugated to each antibody molecule. It is crucial for optimizing the therapeutic efficacy and safety profile of the ADC. Contact us for more detailed reference data.
Our service provides precise measurement of target antigen levels on cell surfaces, crucial for identifying and validating new ADC targets. Utilizing advanced flow cytometry, we quantify antigen density on various cell types, ensuring target suitability for ADC development. This detailed analysis supports the selection of high-value targets, enhancing the specificity and efficacy of next-generation cancer therapies. Contact us for more detailed reference data.
SPR provides detailed analysis of the binding interactions between therapeutic antibodies and their target antigens, which is essential for designing effective ADCs. The affinity of ADC should be similar as that of naked antibody. Contact us for more detailed reference data.
Flow cytometry is a powerful technique for analyzing the binding of antibodies to target antigens, on the surface of different cell lines. This service is essential for evaluating the binding affinity and specificity of antibodies and ADCs across various cell lines. The data is essential for optimizing ADC development, ensuring targeted delivery to specific cancer cells, and minimizing off-target effects. Contact us for more detailed reference data.
Our Internalization Assay Service utilizes Incucyte live-cell imaging, flow cytometry, and high-content analysis to provide detailed, real-time analysis of ADC internalization into target cancer cells. By combining these advanced technologies, we offer precise, quantitative insights into the uptake and intracellular trafficking of fluorescently labeled ADCs, ensuring optimal design and efficacy of your ADC therapies. Contact us for more detailed reference data.
1. Incucyte provides real-time, continuous monitoring of ADC internalization in live cells. This system captures dynamic processes, allowing visualization of ADC internalization and intracellular trafficking over extended periods, which helps in understanding the temporal aspects of ADC behavior.
2. Temperature shift-based internalization assay is a classical method to evaluate the antibody internalization rate, by calculating MFI of the surface antibody in two groups at 37°C and 4°C respectively.
3. The internalization efficiency of the antibody can be assessed by measuring the cell killing effect, by using Toxin-based cytotoxicity assay.
4. The pH-indicator based internalization flow cytometry assay provides a specific and accurate method for measuring ADC internalization. By utilizing pH-sensitive dyes, this technique ensures that only ADCs internalized into the acidic compartments of cells are detected, offering precise insights into the efficiency and kinetics of ADC internalization and trafficking.
5. High-Content Analysis combines automated microscopy and image analysis to provide detailed, multiparametric data on ADC internalization at the single-cell level. It allows for in-depth analysis of intracellular localization, colocalization with organelles, and morphological changes, offering comprehensive insights into the mechanisms of ADC internalization.
We value your inquiries and are here to provide you with tailored solutions for your drug discovery and development needs. Whether you have questions, require more information, or are interested in discussing potential collaborations, our team of experts is just a message away.
Feel free to reach out to us.