This note extends our previous application note on biochemical characterization of an AR–BRD4 RIPTAC system. Here, we add a structure-guided layer to the story by examining II-5 through docking, molecular dynamics (MD), and ternary-complex modeling.
The data support several practical points: II-5 shows similar predicted binding modes in BRD4-BD1 and BRD4-BD2; its inactive stereochemical variant fits poorly in the bromodomain pocket; AR binding is retained while agonist-like activity appears limited; and among two modeled ternary conformations, pose 2 is the more stable working model.
These results should be interpreted as predictive structural analysis rather than stand-alone mechanistic proof. For RIPTAC programs, this type of in silico work is most useful when integrated with biochemical, biophysical, and cellular validation.
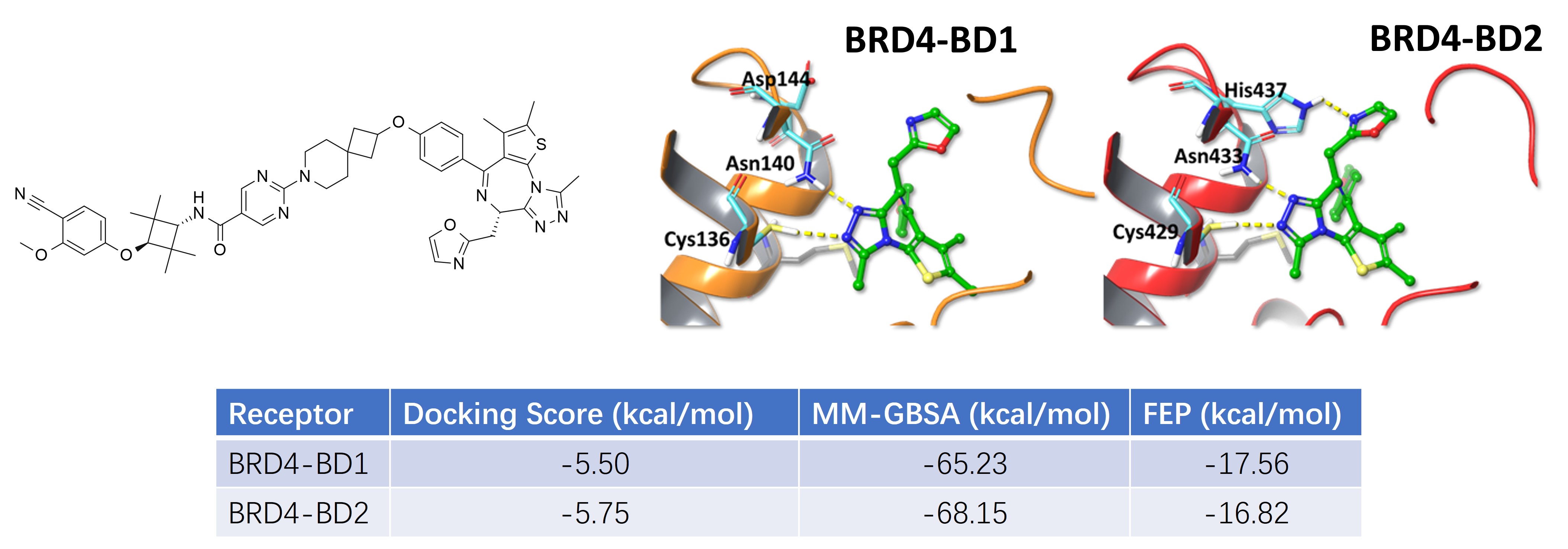
Docking and MD analysis suggest that II-5 adopts a similar binding mode in both BRD4-BD1 and BRD4-BD2. The predicted interaction patterns are broadly consistent across the two bromodomains, and the calculated binding energies are also close.
In the current model set, the docking score, MM-GBSA, and FEP values all support the same conclusion: II-5 can be accommodated by both BD1 and BD2, rather than showing a strong structural bias toward only one bromodomain.
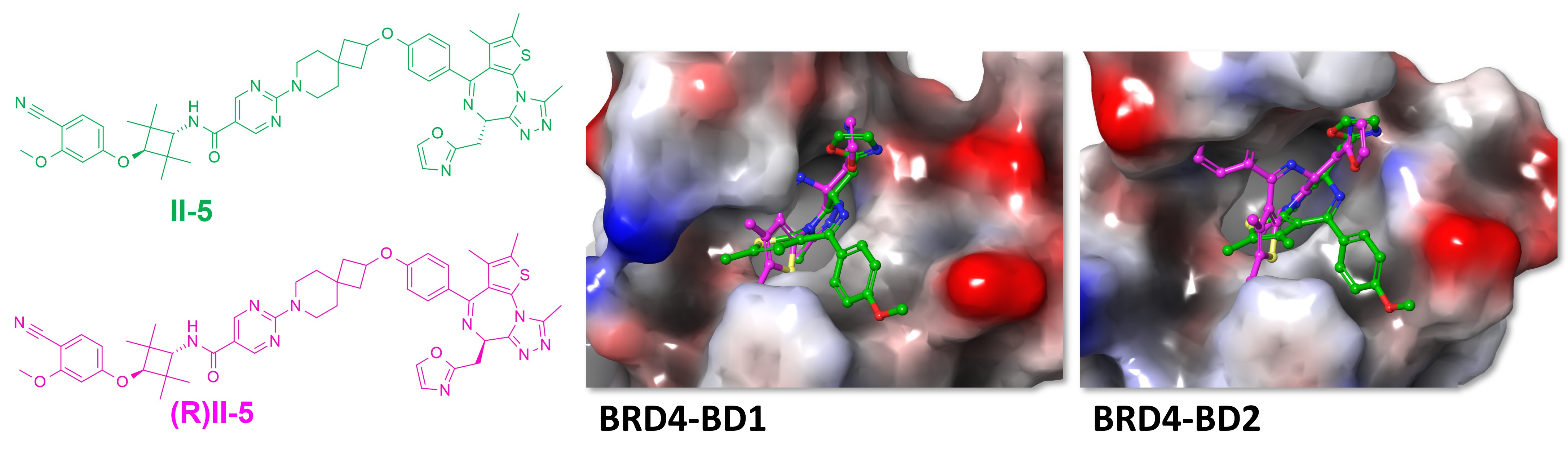
The comparison between the two isomers of II-5 provides a useful structural control. In the docking models, the R-isomer does not fit well into the bromodomain pocket and shows clear steric clashes with the protein environment.
This supports a structure-dependent interpretation: the signal is not simply driven by general occupancy, but by a specific three-dimensional presentation that is compatible with BRD4 pocket geometry.
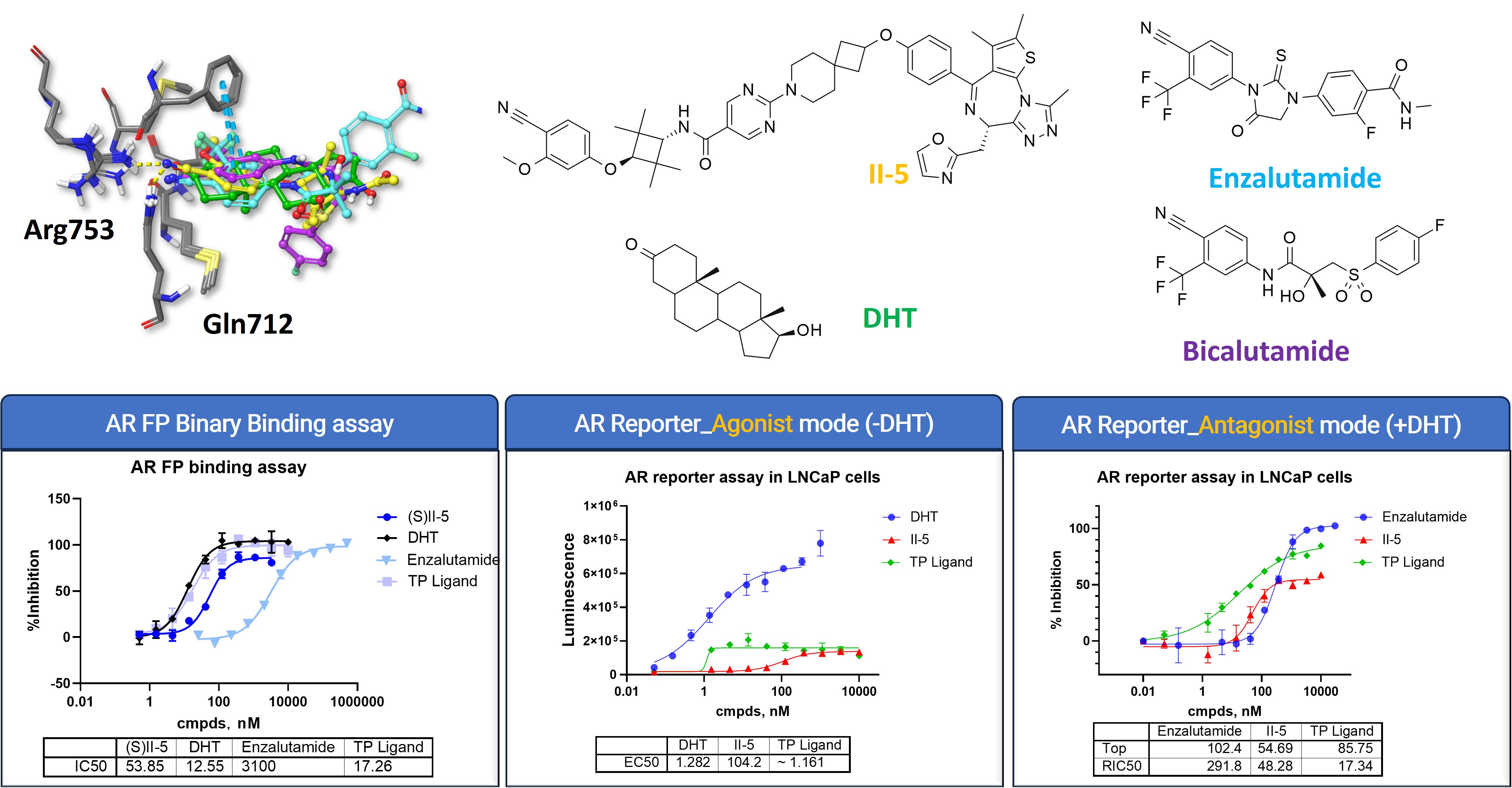
On the AR side, the data indicate that II-5 retains meaningful AR-LBD engagement. At the same time, the reporter-based readouts place its functional profile below that of a strong canonical agonist, supporting a weaker agonist-like behavior in this assay context.
The compounds shown in the figure help frame that interpretation. DHT provides the strong agonist benchmark, while Enzalutamide provides familiar AR antagonist reference. The TP ligand serves as the AR-binding ligand reference within this RIPTAC design context. With these comparators in place, the AR FP binding assay can be read as a binding reference, while the agonist- and antagonist-mode reporter assays help show where II-5 sits functionally relative to established AR ligands.
This distinction matters for AR-centered RIPTAC design, because AR recruitment and downstream AR signaling do not carry the same meaning. A molecule may preserve the target engagement needed for induced proximity while avoiding the stronger agonist-like profile associated with a classical androgen control.
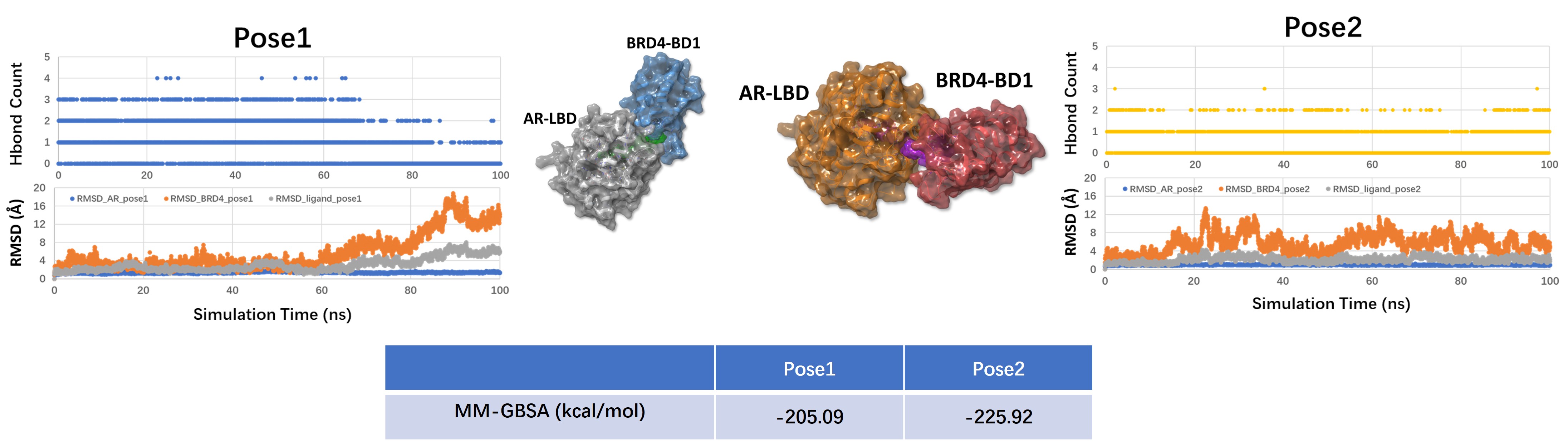
To move beyond binary interactions, we modeled the AR-LBD : II-5 : BRD4-BD1 ternary system. The analysis produced two candidate poses, both of which appeared structurally plausible at the docking stage.
At this point, the question is not whether two poses can be generated, but which pose remains more stable after dynamic refinement and energy analysis.
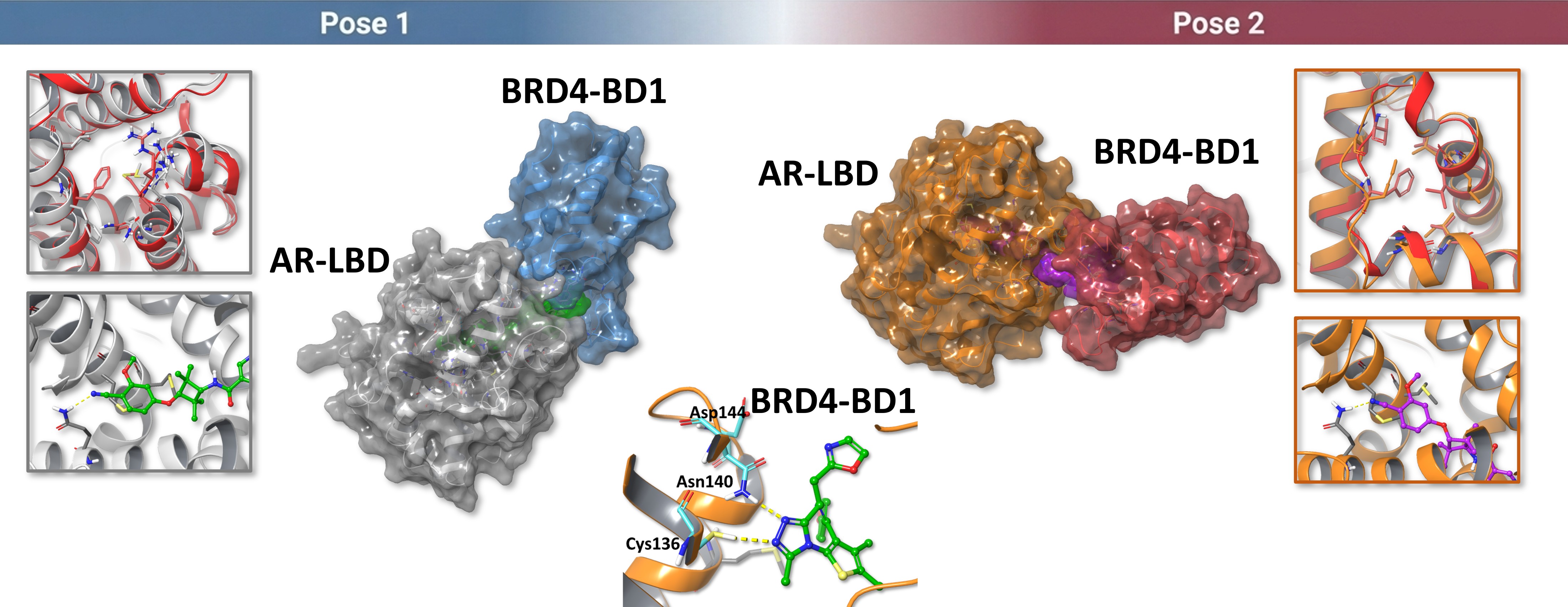
MD trajectory analysis and MM-GBSA calculations support pose 2 as the more stable ternary hypothesis. In the current dataset, pose 2 shows a more favorable MM-GBSA value than pose 1.
This does not prove that pose 2 is the biologically dominant state, but it gives a more credible structural basis for downstream interpretation and follow-up validation.
Based on the preferred ternary pose, the model suggests that II-5 may support a neoPPI-like interface between AR-LBD and BRD4-BD1.
This is relevant for RIPTAC interpretation because productive induced proximity requires more than dual binding. It requires a geometry that can plausibly organize the two proteins into a functionally meaningful arrangement.
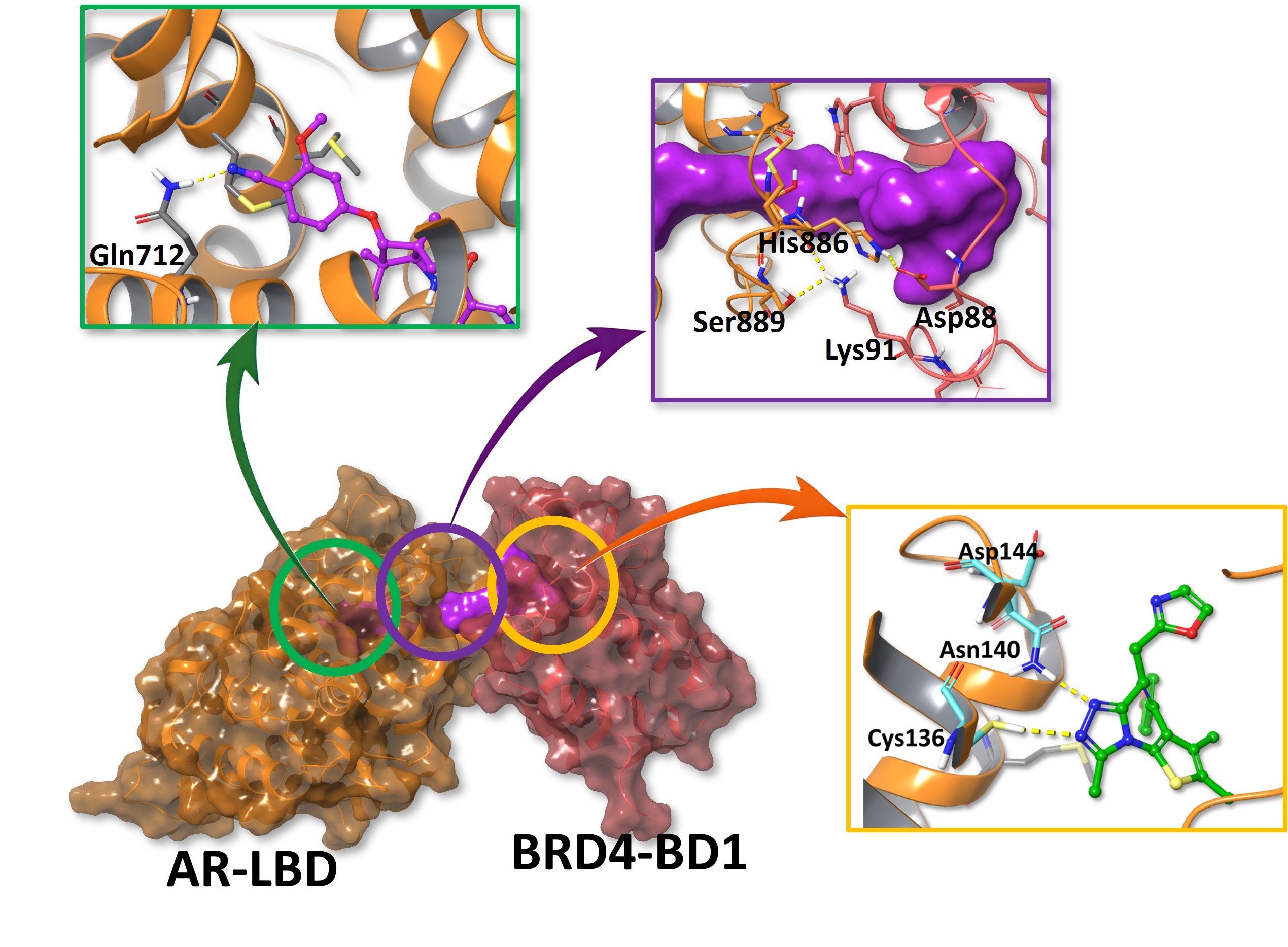
In this case, in silico analysis adds useful structure-guided context to the biochemical characterization workflow. The data support similar BRD4 bromodomain accommodation, clear stereochemical dependence, retained AR engagement, and a preferred ternary pose with a plausible neoPPI-like interface.
These results are predictive and should be interpreted together with orthogonal validation rather than as mechanistic proof on their own.
PREV:From Binary Binding to Functional Readou...
NEXT:There's no more
2026-05-12
2025-10-30
2025-10-23
2025-09-28
We value your inquiries and are here to provide you with tailored solutions for your drug discovery and development needs. Whether you have questions, require more information, or are interested in discussing potential collaborations, our team of experts is just a message away.
Feel free to reach out to us.